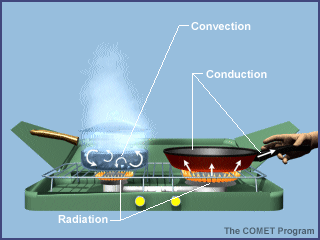
Conduction
~ The transfer of thermal energy with no overall transfer of matter is called conduction.
~ Conduction in gases is slower than in liquids and solids because the particles in a gas collide less often.
~ Conduction is faster in metals than in other solids because metals have free electrons that transfer thermal heat.
~ Thermal energy is transferred without transfer of matter.
~ In most solids, conduction takes place as particles vibrate in place.
~ Thermal conductors conduct energy well. Some examples of thermal conductors are copper and aluminum.
~ Thermal insulators conduct energy poorly. Some examples of thermal insulators are wood and plastic foam.
Convection
~ The tranfer of thermal energy when particles of a fluid move from one place to another is called convection.
~ When air at the bottom of the oven heats up, it expands and becomes less dense than the surrounding air.
Like an oven when you cook food (example at the bottom of page).
~ When a fluid circulates in a loop as it alternately heats up and cools down a convection current occurs.
~ Some examples of convection currents are ocean currents, weather systems, and movement of hot rocks in Earth's
interior.
Radiation
~ The transfer of energy by waves moving through space is called radiation.
~ Energy is transferred by waves.
~ All object radiate energy
~ The farther away you are from a radiating object, the less radiation you recieve.
Thermodynamics
~ Thermodynamics is the study of conversions between thermal energy and other forms of energy.
~ Energy cannot be created or destroyed, but it can be converted into different forms.
~ Thermal energy follows spontaneously from hotter objects to colder ones.
~ The first law of thermodynamics states that energy is conserved.
~ The second law of thermodynamics states that thermal energy can follow from colder object to hotter objects
only if work is done on the system.
~ Thermal energy that is not converted into work is called waste heat.
~ Matter cannot be cooled to absolute zero.
~ Conduction in gases is slower than in liquids and solids because the particles in a gas collide less often.
~ Conduction is faster in metals than in other solids because metals have free electrons that transfer thermal heat.
~ Thermal energy is transferred without transfer of matter.
~ In most solids, conduction takes place as particles vibrate in place.
~ Thermal conductors conduct energy well. Some examples of thermal conductors are copper and aluminum.
~ Thermal insulators conduct energy poorly. Some examples of thermal insulators are wood and plastic foam.
Convection
~ The tranfer of thermal energy when particles of a fluid move from one place to another is called convection.
~ When air at the bottom of the oven heats up, it expands and becomes less dense than the surrounding air.
Like an oven when you cook food (example at the bottom of page).
~ When a fluid circulates in a loop as it alternately heats up and cools down a convection current occurs.
~ Some examples of convection currents are ocean currents, weather systems, and movement of hot rocks in Earth's
interior.
Radiation
~ The transfer of energy by waves moving through space is called radiation.
~ Energy is transferred by waves.
~ All object radiate energy
~ The farther away you are from a radiating object, the less radiation you recieve.
Thermodynamics
~ Thermodynamics is the study of conversions between thermal energy and other forms of energy.
~ Energy cannot be created or destroyed, but it can be converted into different forms.
~ Thermal energy follows spontaneously from hotter objects to colder ones.
~ The first law of thermodynamics states that energy is conserved.
~ The second law of thermodynamics states that thermal energy can follow from colder object to hotter objects
only if work is done on the system.
~ Thermal energy that is not converted into work is called waste heat.
~ Matter cannot be cooled to absolute zero.